
The chem/physics section of the MCAT is scored 118-132 on a curved scale, with the median score of all test takers set to be 125. These discrete questions test both your science knowledge and application of that knowledge based on these four skills, although they tend more toward Skill 1. You will also be asked 15 discrete questions that are completely separate from the ten passages. The questions will address the four Scientific and Reasoning Skills listed, although different passages will focus on different skills. When the incoming nucleophile in an acyl substitution is a water molecule, the reaction is also referred to as an acyl hydrolysis.The MCAT will present you with ten passages based on chemistry and physics subjects, and then present four to seven questions about each passage. For this reason, nucleophilic acyl substitutions are also commonly referred to as acyl transfer reactions. For example, in the reaction below, one alcohol X group (3-methyl-1-butanol) is replaced by another alcohol X group (methanol), as one ester is converted to another.Īnother way of looking at this reaction is to picture the acyl group being transferred from one acyl X group to another: in the example above, the acetyl group is being transferred from 3-methyl-1-butanol to methanol. This is why this reaction type is called a nucleophilic acyl substitution: one acyl X group is substituted for another. Notice that in the product, the nucleophile becomes the new acyl X group. The tetrahedral intermediate rapidly collapses: the carbon-oxygen double bond re-forms, and the acyl X group is expelled. What this means is that the tetrahedral product formed from attack of the nucleophile on the carbonyl carbon is not the product: it is a reactive intermediate. In carboxylic acid derivatives, the acyl X group is a potential leaving group. Just like in aldehydes and ketones, carboxylic acid derivatives are attacked from one side of their trigonal planar carbonyl carbon by a nucleophile, converting this carbon to tetrahedral (sp 3) geometry. The fact that the atom adjacent to the carbonyl carbon in carboxylic acid derivatives is an electronegative heteroatom – rather than a carbon like in ketones or a hydrogen like in aldehydes - is critical to understanding the reactivity of these functional groups. The nucleophilic acyl substitution reaction Notice that the acyl X groups are simply deprotonated forms of other functional groups: in an amide, for example, the acyl X group is an amine, while in an ester the acyl X group is an alcohol.  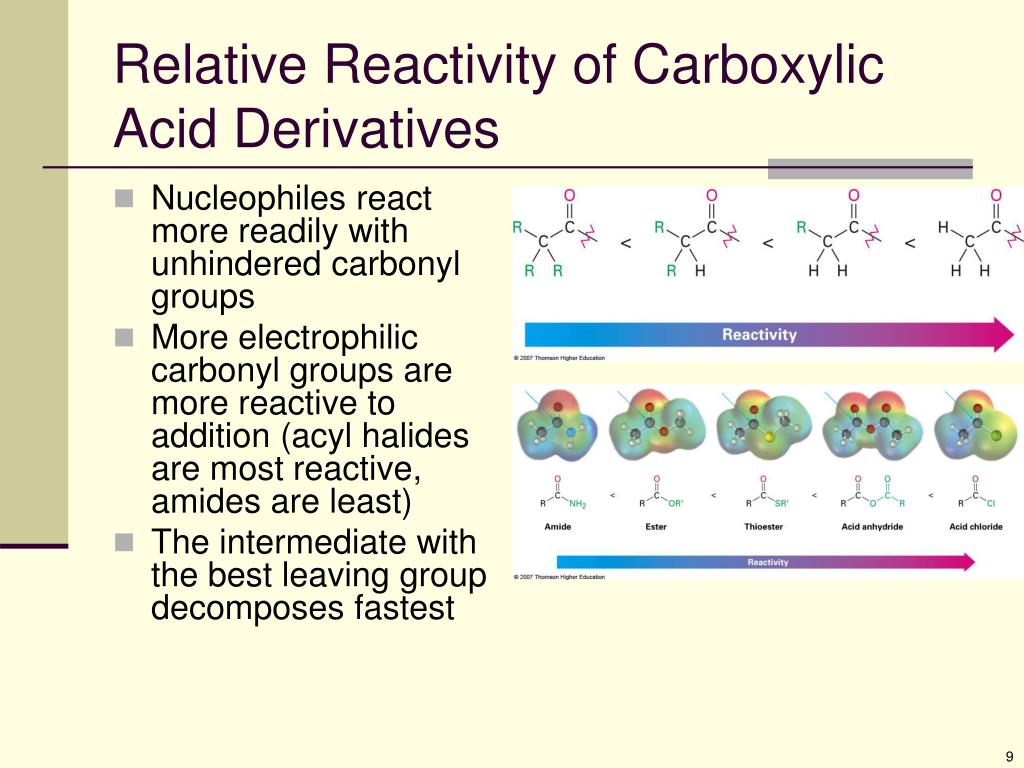
One the other side is the heteroatom-containing group: in this text, we will sometimes refer to this component as the ‘acyl X' group (this, however, is not a standard term in organic chemistry). One side is the carbonyl group and the attached alkyl group: this is called an acyl group (in the specific case where R is a methyl group, the term acetyl group is used). You can think of a carboxylic acid derivative as having two sides. The carboxylic acid derivatives can be distinguished from aldehydes and ketones by the presence of a group containing an electronegative heteroatom - usually oxygen, nitrogen, or sulfur – bonded directly to the carbonyl carbon. Two more examples of carboxylic acid derivatives which are less biologically relevant but important in laboratory synthesis are carboxylic acid anyhydrides and acid chlorides. The functional groups that undergo nucleophilic acyl substitutions are called carboxylic acid derivatives: these include carboxylic acids themselves, carboxylates (deprotonated carboxylic acids), amides, esters, thioesters, and acyl phosphates. \)Ĭarboxylic acid derivatives and acyl groups
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
